The other chalcogenides, ZnSe and ZnTe, have applications in electronics and optics. bell tent sewing pattern; high low passing concepts; are volunteer fire departments government entities; In another example of acidic silicon dioxide reacting with a base, the Blast Furnace extraction of iron, calcium oxide from limestone reacts with silicon dioxide to produce a liquid slag, calcium silicate: Two phosphorus oxides, phosphorus(III) oxide, P4O6, and phosphorus(V) oxide, P4O10, are considered here. WebZinc Oxide | OZn | CID 14806 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety/hazards/toxicity information, supplier lists, and more. Superoxides: Often Potassium, Rubidium, and Cesium react with excess oxygen to produce the superoxide, \( MO_2 \). When HSO 4 - interacts with water, for example, it produces both hydroxide and hydronium ions:. A base is a substance that is used to neutralize acid is called a base. A reasonably concentrated solution of sulfurous acid has a pH of about 1. however, since hydroxides contain close to half as many electrons (hydrogen atoms) per ionic charge as oxygens do, even weak bases can outnumber strong acids due to their ability to absorb additional protons/electrons from other sources at equivalently lower energy cost required to produce hydroxides. Whilst calcium is somewhat larger than magnesium, there is a steady decrease in size as atomic number increases from calcium to zinc. Only one acid is commonly considered, phosphoric(V) acid, H3PO4 (also known as phosphoric acid or as orthophosphoric acid). This means that an Alkaline substance will have a higher concentration of Hydrogen ions compared to another compound.
 Are alkali and base the same? An oxide that can react chemically as either an acid or a base is known as an amphoteric oxide. 8 Which is more acidic zinc sulphate or zinc hydroxide? Chlorine(VII) oxide: Chlorine(VII) oxide is the highest oxide of chlorinethe chlorine atom is in its maximum oxidation state of +7. It adopts a polymeric structure consisting of tetrahedral zinc centres linked by bridging cyanide ligands. The classification of oxides is done into neutral, amphoteric and basic or acidic based on their acid-base characteristics. Alkyl and aryl zinc compounds are contain the linear CZnC motif. Examples of bases: zinc hydroxide and copper oxide. PbO2reacts with hydrochloric acid so, it is basic. Reaction with water: Aluminum oxide is insoluble in water and does not react like sodium oxide and magnesium oxide. Cl2O, SO2, P4O10). This reaction runs essentially to completion: \[ H_2SO_4 (aq) + H_2O (l) \rightarrow H_3P^+ + HSO_4^- (aq)\]. For example, in third period, the behavior of oxides changes as follows: \(\underset{\large{Basic}}{\underbrace{Na_2O,\: MgO}}\hspace{20px} In fact the low-molecular weight compounds will ignite spontaneously on contact with air and are immediately destroyed by reaction with water molecules.
Are alkali and base the same? An oxide that can react chemically as either an acid or a base is known as an amphoteric oxide. 8 Which is more acidic zinc sulphate or zinc hydroxide? Chlorine(VII) oxide: Chlorine(VII) oxide is the highest oxide of chlorinethe chlorine atom is in its maximum oxidation state of +7. It adopts a polymeric structure consisting of tetrahedral zinc centres linked by bridging cyanide ligands. The classification of oxides is done into neutral, amphoteric and basic or acidic based on their acid-base characteristics. Alkyl and aryl zinc compounds are contain the linear CZnC motif. Examples of bases: zinc hydroxide and copper oxide. PbO2reacts with hydrochloric acid so, it is basic. Reaction with water: Aluminum oxide is insoluble in water and does not react like sodium oxide and magnesium oxide. Cl2O, SO2, P4O10). This reaction runs essentially to completion: \[ H_2SO_4 (aq) + H_2O (l) \rightarrow H_3P^+ + HSO_4^- (aq)\]. For example, in third period, the behavior of oxides changes as follows: \(\underset{\large{Basic}}{\underbrace{Na_2O,\: MgO}}\hspace{20px} In fact the low-molecular weight compounds will ignite spontaneously on contact with air and are immediately destroyed by reaction with water molecules. 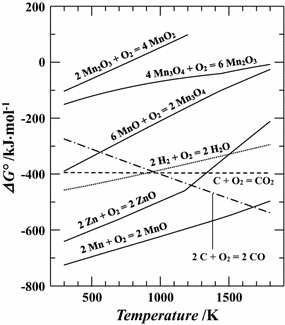 They react with acids to produce salts, e.g., \[ \ce{ MgO + 2HCl \rightarrow MgCl_2 + H_2O } \label{9}\], \[ \ce{ Na_2O + H_2SO_4 \rightarrow Na_2SO_4 + H_2O} \label{10}\]. Zinc sulfide, ZnS, crystallizes in two closely related structures, the zincblende crystal structure and the Wurtzite crystal structure, which are common structures of compounds with the formula MA.
They react with acids to produce salts, e.g., \[ \ce{ MgO + 2HCl \rightarrow MgCl_2 + H_2O } \label{9}\], \[ \ce{ Na_2O + H_2SO_4 \rightarrow Na_2SO_4 + H_2O} \label{10}\]. Zinc sulfide, ZnS, crystallizes in two closely related structures, the zincblende crystal structure and the Wurtzite crystal structure, which are common structures of compounds with the formula MA.  National Center for Biotechnology Information. If soluble in water, they react with water to produce hydroxides (alkalies) e.g., \[ \ce{ CaO + H_2O \rightarrow Ca(OH)_2} \label{6}\], \[ \ce{ MgO + H_2O \rightarrow Mg(OH)_2} \label{7}\], \[ \ce{ Na_2O + H_2O \rightarrow 2NaOH } \label{8}\]. Accessibility StatementFor more information contact us atinfo@libretexts.orgor check out our status page at https://status.libretexts.org.
National Center for Biotechnology Information. If soluble in water, they react with water to produce hydroxides (alkalies) e.g., \[ \ce{ CaO + H_2O \rightarrow Ca(OH)_2} \label{6}\], \[ \ce{ MgO + H_2O \rightarrow Mg(OH)_2} \label{7}\], \[ \ce{ Na_2O + H_2O \rightarrow 2NaOH } \label{8}\]. Accessibility StatementFor more information contact us atinfo@libretexts.orgor check out our status page at https://status.libretexts.org. With the oyxgen exhibiting an oxidation number of -2. For zinc the pH value was about 5.8, for nickel 6.3, and for copper 4.5.
 Zinc oxide is a base, because it reacts with an acid to form a salt and water. the Zn-ligand bonds exchange with other ligands rapidly. By direct heating of an element with oxygen: Many metals and non-metals burn rapidly when heated in oxygen or air, producing their oxides, e.g., \[P_4 + 5O_2 \xrightarrow{Heat} 2P_2O_5\]. For example, terpyridine, which is a tridentate ligand forms the complex [Zn(terpy)Cl2]. \[Al_2O_3 + 6HCl \rightarrow 2AlCl_3 + 3H_2O\]. eduardo franco turbotax commercial spanish.
Zinc oxide is a base, because it reacts with an acid to form a salt and water. the Zn-ligand bonds exchange with other ligands rapidly. By direct heating of an element with oxygen: Many metals and non-metals burn rapidly when heated in oxygen or air, producing their oxides, e.g., \[P_4 + 5O_2 \xrightarrow{Heat} 2P_2O_5\]. For example, terpyridine, which is a tridentate ligand forms the complex [Zn(terpy)Cl2]. \[Al_2O_3 + 6HCl \rightarrow 2AlCl_3 + 3H_2O\]. eduardo franco turbotax commercial spanish. www.transtutors.com/chemistryts/oxides.aspx. An amphoteric solution is a substance that can chemically react as either acid or base. Is the salt zinc acidic or basic in nature? What are the products of metal oxide with an acid? Oxides can be generated via multiple reactions. Oxygen can thus be obtained from acidified water by its electrolysis. National Library of Medicine. In the second case (using twice as much sodium hydroxide), both protons react. Aluminum oxide reacts with hot, concentrated sodium hydroxide solution to produce a colorless solution of sodium tetrahydroxoaluminate: \[Al_2O_3 + 2NaOH +3H_2O \rightarrow 2NaAl(OH)_4\]. Reaction with acids: As a strong base, sodium oxide also reacts with acids. It is basic because it contains the oxide ion, O 2-, which is a very strong base with a high tendency to combine with hydrogen ions. Zinc hydroxide, Zn(OH)2 is also amphoteric. is proficient a good score on indeed. [2] It crystallizes with the Wurtzite structure. Zinc oxide is perhaps best known as an ingredient in cosmetics, personal care products, medicines, and sunscreens. This reaction is more appropriately described as an equilibrium: \[ HSO_4^- (aq) + H_2O \rightleftharpoons H_3O^+ (aq) + SO_4^{2-} (aq)\]. The Europe market for Zinc Oxide Sunscreen is estimated to increase from USD million in 2022 to reach USD million by 2030, at a CAGR of Percent during the forecast period of 2023 through 2030. Enjoy learning with a great experience. Necessary cookies are absolutely essential for the website to function properly. Zinc Oxide is an Amphoteric Oxide (a molecule or ion that can react both as an acid as well as a base is called amphoteric. It continues the trend of the highest oxides of the Period 3 elements towards being stronger acids. They are called amphoteric oxides.

 These are termed as oxides because here, oxygen is in combination with only one element. But opting out of some of these cookies may affect your browsing experience. It has reactions as both a base and an acid. By clicking Accept All, you consent to the use of ALL the cookies. Zn2+ is a class A acceptor in the classification of Ahrland, Chatt and Davies,[16] and so forms stronger complexes with the first-row donor atoms oxygen or nitrogen than with second-row sulfur or phosphorus. Find notes, question papers for other subjects like Mathematics, Physics, Biology and various competitive exams as well. WebZinc Oxide | OZn | CID 14806 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety/hazards/toxicity information, supplier lists, and more. National Center for Biotechnology Information. The base is bitter in taste and is slippery in nature. Watch the video to better understand the difference between alkali and bases. The reaction is shown below: Reaction with acids: Magnesium oxide reacts with acids as predicted for a simple metal oxide. Sulfuric acid displays all the reactions characteristic of a strong acid. Alkalis are a subset of bases, but all alkalis are bases. In fact, the hydrogen sulfate ion is a relatively weak acid, similar in strength to the acids discussed above. HSO 4-+H 2 OSO 2 4-+H 3 O +; HSO 4 - +H 2 OH 2 SO 4 +OH ; Both basic and acidic characteristics are found in amphoteric The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The Crystal Structure and Molecular Stereochemistry of Tetra(4-pyridyl)porphinatomonopyridinezinc(II). Tetrahedrally coordinated zinc is found in metallo-enzymes such as carbonic anhydrase. This means that these substances dissolve easily in water. ZnO reacts with hydrochloric acid forming zinc chloride and water. 4. is proficient a good score on indeed. However, it acts as a basic oxide while reacting with hydrochloric acid. Neutral chloric(VII) acid has the following structure: When the chlorate(VII) ion (perchlorate ion) forms by loss of a proton (in a reaction with water, for example), the charge is delocalized over every oxygen atom in the ion. NCERT Solutions Class 12 Business Studies, NCERT Solutions Class 12 Accountancy Part 1, NCERT Solutions Class 12 Accountancy Part 2, NCERT Solutions Class 11 Business Studies, NCERT Solutions for Class 10 Social Science, NCERT Solutions for Class 10 Maths Chapter 1, NCERT Solutions for Class 10 Maths Chapter 2, NCERT Solutions for Class 10 Maths Chapter 3, NCERT Solutions for Class 10 Maths Chapter 4, NCERT Solutions for Class 10 Maths Chapter 5, NCERT Solutions for Class 10 Maths Chapter 6, NCERT Solutions for Class 10 Maths Chapter 7, NCERT Solutions for Class 10 Maths Chapter 8, NCERT Solutions for Class 10 Maths Chapter 9, NCERT Solutions for Class 10 Maths Chapter 10, NCERT Solutions for Class 10 Maths Chapter 11, NCERT Solutions for Class 10 Maths Chapter 12, NCERT Solutions for Class 10 Maths Chapter 13, NCERT Solutions for Class 10 Maths Chapter 14, NCERT Solutions for Class 10 Maths Chapter 15, NCERT Solutions for Class 10 Science Chapter 1, NCERT Solutions for Class 10 Science Chapter 2, NCERT Solutions for Class 10 Science Chapter 3, NCERT Solutions for Class 10 Science Chapter 4, NCERT Solutions for Class 10 Science Chapter 5, NCERT Solutions for Class 10 Science Chapter 6, NCERT Solutions for Class 10 Science Chapter 7, NCERT Solutions for Class 10 Science Chapter 8, NCERT Solutions for Class 10 Science Chapter 9, NCERT Solutions for Class 10 Science Chapter 10, NCERT Solutions for Class 10 Science Chapter 11, NCERT Solutions for Class 10 Science Chapter 12, NCERT Solutions for Class 10 Science Chapter 13, NCERT Solutions for Class 10 Science Chapter 14, NCERT Solutions for Class 10 Science Chapter 15, NCERT Solutions for Class 10 Science Chapter 16, NCERT Solutions For Class 9 Social Science, NCERT Solutions For Class 9 Maths Chapter 1, NCERT Solutions For Class 9 Maths Chapter 2, NCERT Solutions For Class 9 Maths Chapter 3, NCERT Solutions For Class 9 Maths Chapter 4, NCERT Solutions For Class 9 Maths Chapter 5, NCERT Solutions For Class 9 Maths Chapter 6, NCERT Solutions For Class 9 Maths Chapter 7, NCERT Solutions For Class 9 Maths Chapter 8, NCERT Solutions For Class 9 Maths Chapter 9, NCERT Solutions For Class 9 Maths Chapter 10, NCERT Solutions For Class 9 Maths Chapter 11, NCERT Solutions For Class 9 Maths Chapter 12, NCERT Solutions For Class 9 Maths Chapter 13, NCERT Solutions For Class 9 Maths Chapter 14, NCERT Solutions For Class 9 Maths Chapter 15, NCERT Solutions for Class 9 Science Chapter 1, NCERT Solutions for Class 9 Science Chapter 2, NCERT Solutions for Class 9 Science Chapter 3, NCERT Solutions for Class 9 Science Chapter 4, NCERT Solutions for Class 9 Science Chapter 5, NCERT Solutions for Class 9 Science Chapter 6, NCERT Solutions for Class 9 Science Chapter 7, NCERT Solutions for Class 9 Science Chapter 8, NCERT Solutions for Class 9 Science Chapter 9, NCERT Solutions for Class 9 Science Chapter 10, NCERT Solutions for Class 9 Science Chapter 11, NCERT Solutions for Class 9 Science Chapter 12, NCERT Solutions for Class 9 Science Chapter 13, NCERT Solutions for Class 9 Science Chapter 14, NCERT Solutions for Class 9 Science Chapter 15, NCERT Solutions for Class 8 Social Science, NCERT Solutions for Class 7 Social Science, NCERT Solutions For Class 6 Social Science, CBSE Previous Year Question Papers Class 10, CBSE Previous Year Question Papers Class 12, Important Questions For Class 12 Chemistry, Important Questions For Class 11 Chemistry, Important Questions For Class 10 Chemistry, Important Questions For Class 9 Chemistry, Important Questions For Class 8 Chemistry, Important Questions For Class 7 Chemistry, Important Questions For Class 6 Chemistry, Class 12 Chemistry Viva Questions With Answers, Class 11 Chemistry Viva Questions With Answers, Class 10 Chemistry Viva Questions With Answers, Class 9 Chemistry Viva Questions With Answers, CBSE Previous Year Question Papers Class 10 Science, CBSE Previous Year Question Papers Class 12 Physics, CBSE Previous Year Question Papers Class 12 Chemistry, CBSE Previous Year Question Papers Class 12 Biology, ICSE Previous Year Question Papers Class 10 Physics, ICSE Previous Year Question Papers Class 10 Chemistry, ICSE Previous Year Question Papers Class 10 Maths, ISC Previous Year Question Papers Class 12 Physics, ISC Previous Year Question Papers Class 12 Chemistry, ISC Previous Year Question Papers Class 12 Biology, JEE Main 2023 Question Papers with Answers, JEE Main 2022 Question Papers with Answers, JEE Advanced 2022 Question Paper with Answers. Legal.
These are termed as oxides because here, oxygen is in combination with only one element. But opting out of some of these cookies may affect your browsing experience. It has reactions as both a base and an acid. By clicking Accept All, you consent to the use of ALL the cookies. Zn2+ is a class A acceptor in the classification of Ahrland, Chatt and Davies,[16] and so forms stronger complexes with the first-row donor atoms oxygen or nitrogen than with second-row sulfur or phosphorus. Find notes, question papers for other subjects like Mathematics, Physics, Biology and various competitive exams as well. WebZinc Oxide | OZn | CID 14806 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety/hazards/toxicity information, supplier lists, and more. National Center for Biotechnology Information. The base is bitter in taste and is slippery in nature. Watch the video to better understand the difference between alkali and bases. The reaction is shown below: Reaction with acids: Magnesium oxide reacts with acids as predicted for a simple metal oxide. Sulfuric acid displays all the reactions characteristic of a strong acid. Alkalis are a subset of bases, but all alkalis are bases. In fact, the hydrogen sulfate ion is a relatively weak acid, similar in strength to the acids discussed above. HSO 4-+H 2 OSO 2 4-+H 3 O +; HSO 4 - +H 2 OH 2 SO 4 +OH ; Both basic and acidic characteristics are found in amphoteric The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The Crystal Structure and Molecular Stereochemistry of Tetra(4-pyridyl)porphinatomonopyridinezinc(II). Tetrahedrally coordinated zinc is found in metallo-enzymes such as carbonic anhydrase. This means that these substances dissolve easily in water. ZnO reacts with hydrochloric acid forming zinc chloride and water. 4. is proficient a good score on indeed. However, it acts as a basic oxide while reacting with hydrochloric acid. Neutral chloric(VII) acid has the following structure: When the chlorate(VII) ion (perchlorate ion) forms by loss of a proton (in a reaction with water, for example), the charge is delocalized over every oxygen atom in the ion. NCERT Solutions Class 12 Business Studies, NCERT Solutions Class 12 Accountancy Part 1, NCERT Solutions Class 12 Accountancy Part 2, NCERT Solutions Class 11 Business Studies, NCERT Solutions for Class 10 Social Science, NCERT Solutions for Class 10 Maths Chapter 1, NCERT Solutions for Class 10 Maths Chapter 2, NCERT Solutions for Class 10 Maths Chapter 3, NCERT Solutions for Class 10 Maths Chapter 4, NCERT Solutions for Class 10 Maths Chapter 5, NCERT Solutions for Class 10 Maths Chapter 6, NCERT Solutions for Class 10 Maths Chapter 7, NCERT Solutions for Class 10 Maths Chapter 8, NCERT Solutions for Class 10 Maths Chapter 9, NCERT Solutions for Class 10 Maths Chapter 10, NCERT Solutions for Class 10 Maths Chapter 11, NCERT Solutions for Class 10 Maths Chapter 12, NCERT Solutions for Class 10 Maths Chapter 13, NCERT Solutions for Class 10 Maths Chapter 14, NCERT Solutions for Class 10 Maths Chapter 15, NCERT Solutions for Class 10 Science Chapter 1, NCERT Solutions for Class 10 Science Chapter 2, NCERT Solutions for Class 10 Science Chapter 3, NCERT Solutions for Class 10 Science Chapter 4, NCERT Solutions for Class 10 Science Chapter 5, NCERT Solutions for Class 10 Science Chapter 6, NCERT Solutions for Class 10 Science Chapter 7, NCERT Solutions for Class 10 Science Chapter 8, NCERT Solutions for Class 10 Science Chapter 9, NCERT Solutions for Class 10 Science Chapter 10, NCERT Solutions for Class 10 Science Chapter 11, NCERT Solutions for Class 10 Science Chapter 12, NCERT Solutions for Class 10 Science Chapter 13, NCERT Solutions for Class 10 Science Chapter 14, NCERT Solutions for Class 10 Science Chapter 15, NCERT Solutions for Class 10 Science Chapter 16, NCERT Solutions For Class 9 Social Science, NCERT Solutions For Class 9 Maths Chapter 1, NCERT Solutions For Class 9 Maths Chapter 2, NCERT Solutions For Class 9 Maths Chapter 3, NCERT Solutions For Class 9 Maths Chapter 4, NCERT Solutions For Class 9 Maths Chapter 5, NCERT Solutions For Class 9 Maths Chapter 6, NCERT Solutions For Class 9 Maths Chapter 7, NCERT Solutions For Class 9 Maths Chapter 8, NCERT Solutions For Class 9 Maths Chapter 9, NCERT Solutions For Class 9 Maths Chapter 10, NCERT Solutions For Class 9 Maths Chapter 11, NCERT Solutions For Class 9 Maths Chapter 12, NCERT Solutions For Class 9 Maths Chapter 13, NCERT Solutions For Class 9 Maths Chapter 14, NCERT Solutions For Class 9 Maths Chapter 15, NCERT Solutions for Class 9 Science Chapter 1, NCERT Solutions for Class 9 Science Chapter 2, NCERT Solutions for Class 9 Science Chapter 3, NCERT Solutions for Class 9 Science Chapter 4, NCERT Solutions for Class 9 Science Chapter 5, NCERT Solutions for Class 9 Science Chapter 6, NCERT Solutions for Class 9 Science Chapter 7, NCERT Solutions for Class 9 Science Chapter 8, NCERT Solutions for Class 9 Science Chapter 9, NCERT Solutions for Class 9 Science Chapter 10, NCERT Solutions for Class 9 Science Chapter 11, NCERT Solutions for Class 9 Science Chapter 12, NCERT Solutions for Class 9 Science Chapter 13, NCERT Solutions for Class 9 Science Chapter 14, NCERT Solutions for Class 9 Science Chapter 15, NCERT Solutions for Class 8 Social Science, NCERT Solutions for Class 7 Social Science, NCERT Solutions For Class 6 Social Science, CBSE Previous Year Question Papers Class 10, CBSE Previous Year Question Papers Class 12, Important Questions For Class 12 Chemistry, Important Questions For Class 11 Chemistry, Important Questions For Class 10 Chemistry, Important Questions For Class 9 Chemistry, Important Questions For Class 8 Chemistry, Important Questions For Class 7 Chemistry, Important Questions For Class 6 Chemistry, Class 12 Chemistry Viva Questions With Answers, Class 11 Chemistry Viva Questions With Answers, Class 10 Chemistry Viva Questions With Answers, Class 9 Chemistry Viva Questions With Answers, CBSE Previous Year Question Papers Class 10 Science, CBSE Previous Year Question Papers Class 12 Physics, CBSE Previous Year Question Papers Class 12 Chemistry, CBSE Previous Year Question Papers Class 12 Biology, ICSE Previous Year Question Papers Class 10 Physics, ICSE Previous Year Question Papers Class 10 Chemistry, ICSE Previous Year Question Papers Class 10 Maths, ISC Previous Year Question Papers Class 12 Physics, ISC Previous Year Question Papers Class 12 Chemistry, ISC Previous Year Question Papers Class 12 Biology, JEE Main 2023 Question Papers with Answers, JEE Main 2022 Question Papers with Answers, JEE Advanced 2022 Question Paper with Answers. Legal. \[ H_2 + O_2 \rightarrow H_2O_2 \label{20}\]. Potassium hydroxide is used in agriculture to make acid soil more alkaline so that plants grow stronger in it, and is also used in alkaline, Ni-Cd, and Ni-MH batteries as the electrolyte. By thermal decomposition of certain compounds like hydroxides. Yes, an alkali is a base. Describing the properties of aluminum oxide can be confusing because it exists in a number of different forms.
The Europe market for Zinc Oxide Sunscreen is estimated to increase from USD million in 2022 to reach USD million by 2030, at a CAGR of Percent during the forecast period of 2023 through 2030. It is basic because it contains the oxide ion, O 2-, which is a very strong base with a high tendency to combine with hydrogen ions. Calculations indicate that a zinc compound with the oxidation state of +4 is unlikely to exist.[7]. tetrahydroxozincate) ion, [Zn(OH) 4 ] 2 . { "Acid-base_Behavior_of_the_Oxides" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.
It is amphoteric , dissolving in acids to give the aqueous Zn 2+ ion and in alkali to give the zincate (a.k.a. A concentrated solution of sodium oxide in water will have pH 14. Put your understanding of this concept to test by answering a few MCQs. The LibreTexts libraries arePowered by NICE CXone Expertand are supported by the Department of Education Open Textbook Pilot Project, the UC Davis Office of the Provost, the UC Davis Library, the California State University Affordable Learning Solutions Program, and Merlot. 7 Is the salt zinc acidic or basic in nature? [20][21] The compound zinc cyanide, Zn(CN)2, is not 2-coordinate. Alkalis are characterized by their ability to neutralize acids and form metal oxides when reacting with water. Zinc may be classified as a post-transition main group element with zinc(II). An alkali is a basic soluble hydroxide of alkali metals or alkaline earth metals. The LibreTexts libraries arePowered by NICE CXone Expertand are supported by the Department of Education Open Textbook Pilot Project, the UC Davis Office of the Provost, the UC Davis Library, the California State University Affordable Learning Solutions Program, and Merlot. It is a chemical compound with mild astringent and antiseptic action which works as a topical protectant. It reacts with both acids and bases to form salts.
 Legal. National Institutes of Health. 4: The fourth difference between alkali and base is that the former has an increased pH level while the latter possesses a decreased pH scale.
Legal. National Institutes of Health. 4: The fourth difference between alkali and base is that the former has an increased pH level while the latter possesses a decreased pH scale. While amphoteric looks cheem, it comes from the Greek word ampho that means both.
 WebZinc oxide, ZnO, is the most important manufactured compound of zinc, with a wide variety of uses. A concentrated solution of sodium oxide in water will have pH 14. However, the pH of the resulting solution is about 9, indicating that hydroxide ions have been produced. When heated with oxygen, compounds containing carbon and hydrogen are oxidized. The zinc acetylacetonate complex, Zn(acac)2 is interesting. It occurs in nature as the mineral zincite. Metal oxides on the left side of the periodic table produce basic solutions in water (e.g. Exceptions occur when the compound contains a coloured anion or ligand.
WebZinc oxide, ZnO, is the most important manufactured compound of zinc, with a wide variety of uses. A concentrated solution of sodium oxide in water will have pH 14. However, the pH of the resulting solution is about 9, indicating that hydroxide ions have been produced. When heated with oxygen, compounds containing carbon and hydrogen are oxidized. The zinc acetylacetonate complex, Zn(acac)2 is interesting. It occurs in nature as the mineral zincite. Metal oxides on the left side of the periodic table produce basic solutions in water (e.g. Exceptions occur when the compound contains a coloured anion or ligand.  Aluminum oxide shows acid and basic properties of an oxide, it is amphoteric. No fluoro complexes are known, but complexes with the other halides and with pseudohalides, [ZnX3] and [ZnX4]2 can be prepared. about 5.8 Reaction with water: Sodium oxide reacts exothermically with cold water to produce sodium hydroxide solution. There are many bases that are insoluble-they are not dissolving in water. This page titled Acid-base Behavior of the Oxides is shared under a CC BY-NC 4.0 license and was authored, remixed, and/or curated by Jim Clark. Li2O). Zinc oxide is insoluble in water. 9th Edition. The cookies is used to store the user consent for the cookies in the category "Necessary". WebZinc oxide (ZnO), Tin (IV)oxide or SnO2, Aluminum oxide (Al2O3) and Beryllium oxide (BeO) are all amphoteric substances, meaning they can react with both acids and bases. tetrahydroxozincate) ion, [Zn(OH) 4 ] 2 . Both Zn and S are tetrahedrally coordinated by the other ion.
Aluminum oxide shows acid and basic properties of an oxide, it is amphoteric. No fluoro complexes are known, but complexes with the other halides and with pseudohalides, [ZnX3] and [ZnX4]2 can be prepared. about 5.8 Reaction with water: Sodium oxide reacts exothermically with cold water to produce sodium hydroxide solution. There are many bases that are insoluble-they are not dissolving in water. This page titled Acid-base Behavior of the Oxides is shared under a CC BY-NC 4.0 license and was authored, remixed, and/or curated by Jim Clark. Li2O). Zinc oxide is insoluble in water. 9th Edition. The cookies is used to store the user consent for the cookies in the category "Necessary". WebZinc oxide (ZnO), Tin (IV)oxide or SnO2, Aluminum oxide (Al2O3) and Beryllium oxide (BeO) are all amphoteric substances, meaning they can react with both acids and bases. tetrahydroxozincate) ion, [Zn(OH) 4 ] 2 . Both Zn and S are tetrahedrally coordinated by the other ion.  As the ligand is bidentate a tetrahedral structure might be expected. The reaction is shown below: The following reactions concern the more reactive forms of the molecule. Other uncategorized cookies are those that are being analyzed and have not been classified into a category as yet.
As the ligand is bidentate a tetrahedral structure might be expected. The reaction is shown below: The following reactions concern the more reactive forms of the molecule. Other uncategorized cookies are those that are being analyzed and have not been classified into a category as yet. \[[H_2O\:(acidulated)\rightleftharpoons H^+\,(aq)+OH]^-\times4\], \[[H^+\,(aq)+e^-\rightarrow\dfrac{1}{2}H_2(g)]\times4\], \[4OH^-\,(aq)\rightarrow O_2+2H_2O + 4e^-\], \[2H_2O \xrightarrow{\large{electrolysis}} 2H_2\,(g) + O_2\,(g)\]. WebThere are multiple definitions of the base. Answer: Zno is an amphoteric oxide,because it can react with both acids and bases. Click Start Quiz to begin! General Chemistry: principles and modern applications. A. [24] In this case all four coordination positions are occupied by the histidine and cysteine residues. Square pyramidal 5-coordinate Zinc is found in Tetra(4-pyridyl)porphinatomonopyridinezinc(II) [19] Solution studies of other 5-coordinate Zinc porphyrins have been reported. WebZinc oxide (ZnO), Tin (IV)oxide or SnO2, Aluminum oxide (Al2O3) and Beryllium oxide (BeO) are all amphoteric substances, meaning they can react with both acids and bases.